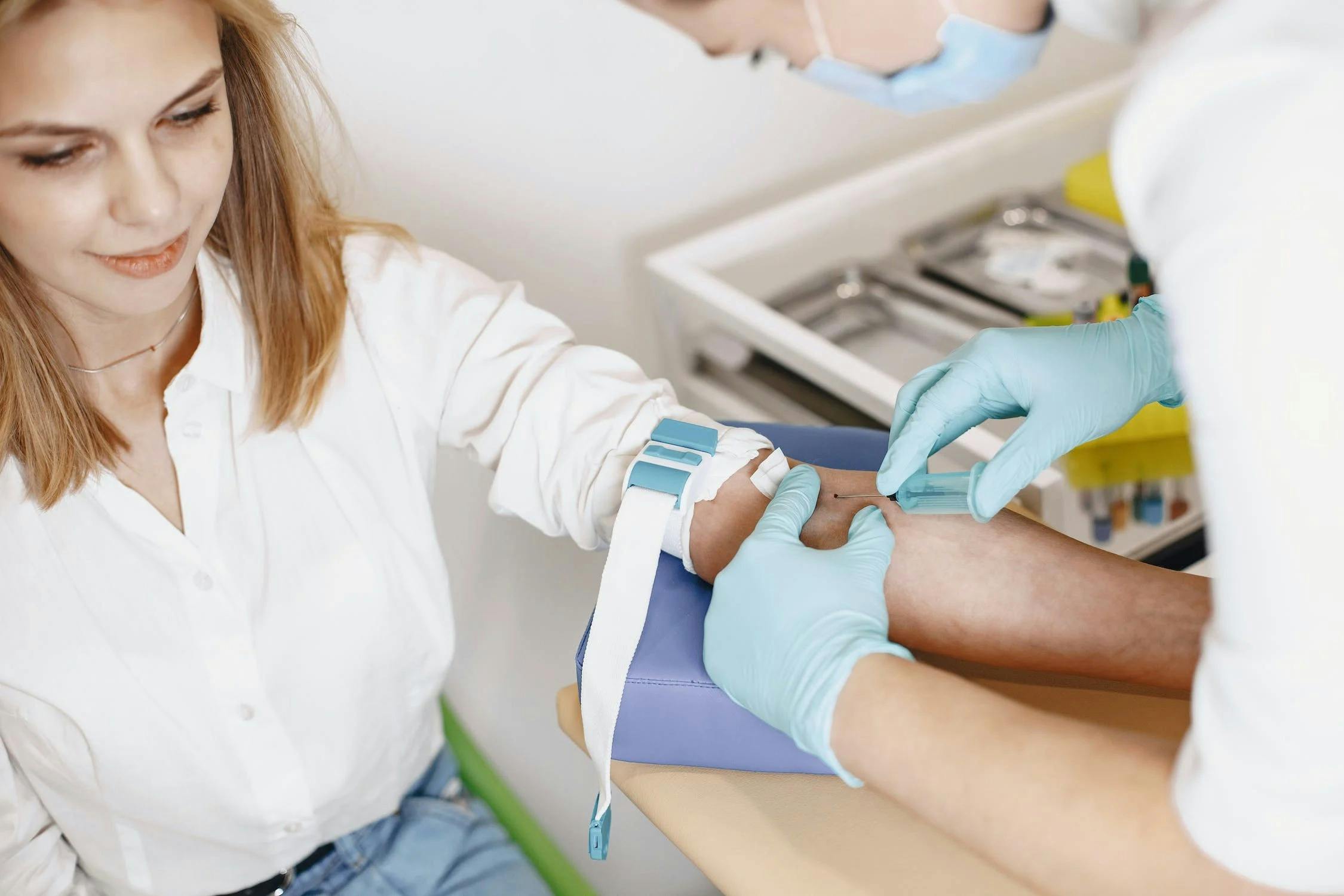
We are proud to announce that we are currently the only medical logistics company in the UK which is accredited by the United Kingdom Accreditation Service (UKAS) and registered with the Care Quality Commission (CQC). This means that our services meet internationally recognised standards - ISO and GMP - and national regulatory requirements for safety, efficacy, and competence.
For us, it's a momentous achievement within the industry and provides validation of the quality and safety of our operations. This accomplishment wouldn't be possible without our amazing team of professionals.
In this blog post we would like to explain more about what it means to be CQC registered and UKAS accredited, and the benefits it brings to us as an organisation, and to our service users.
CQC Registration
CQC is the independent regulator of health and social care in England. They are authorised by the United Kingdom Health Security Agency (UKHSA) to regulate health and social care services and ensure that they provide service users with safe, effective and high-quality care. They also promote a culture of accountability and continuous improvement in the services they monitor. They begin their registration process by performing an initial assessment of the organisation’s Key Lines of Enquiry (KLOEs). This initial assessment determines if the service is safe, effective, caring, responsive and well-led. The regulated service will then undergo an additional assessment which will determine compliance with CQC’s 13 Fundamental Standards - this is derived from The Health and Social Care Act 2008 Regulations 2014 - after which they publish the key findings and performance ratings to help people choose the right care service for them.
Regulation of care services involves:
Although they inspect and regulate different care services in different ways, there are some things that do apply across all services.
Registering organisations that apply to provide services
Using data, evidence and information to assess services
Using feedback service users have given them to help reach judgements
Performing inspections carried out by experts
Publishing information on their judgements and inspections
Taking action when they ascertain that services need to improve
Ensuring those responsible for poor quality care are held accountable for their actions
CQC’s mission is clear. The public should be well informed about the safety and quality of care they can expect from health and social care providers that are registered with them. It’s all about empowering the public and giving them reassurance
The register of Providers and Managers of regulated health and care services helps to ensure transparency regarding who has been registered to provide services in the regulated health and social care market and to provide accountability if things go wrong.
UKAS accreditation
UKAS is appointed under the Accreditation Regulations 2009 as the sole National Accreditation Body for the United Kingdom. Its function is to assess organisations that provide conformity assessment services such as certification, testing, inspection, calibration and verification, against nationally and internationally agreed standards, UKAS is a signatory to European and International bodies recognising UKAS accredited organisations.
More and more businesses are choosing the UKAS accreditation route because of the benefits it brings.
Accreditation can:
Improve organisational efficiency by highlighting gaps or weaknesses in operational capability, thereby providing the opportunity for improved organisational efficiency and outputs
Provide impartial assurance of the technical competence of an organisation delivering confidence in the service provided
Facilitate risk management by independently assessing identifying and reducing risks
Creates confidence in industry supply chains which is often required by public and private sector organisations as a mandatory prerequisite for acceptance
Increase international acceptance for import & export
As of the 23rd of August 2022, we are CQC registered to provide Diagnostic and Screening services (such as phlebotomy and other mobile nursing services). You can find the link to our registration here:https://www.cqc.org.uk/provider/1-10972391656/registration-info#RA8
This summer we also successfully completed our UKAS surveillance assessment, you can access our UKAS accreditation details here: https://www.ukas.com/wp-content/uploads/schedule_uploads/971748/22387-Medical-Multiple.pdf